
Implanting innovation
Leverage the latest innovations to reduce costs, advance quality and extend your production capacity.

Orthopedic Implant Manufacturing
Leverage our agility, ingenuity, automation and low-cost base to drive down your cost of manufacture.

Additive Manufacturing
The expertise, technologies, and scale to deliver end-to-end, world-class, additive manufacturing solutions.

Medical Component Manufacturing
Leverage precise, state-of-the-art Swiss turning technologies to manufacture high quality, complex components.
Capabilities
Joint Partnership
We help drive progress for our medical device customers by enhancing both their production capacity and their product and process innovation.
Manufacturing Capabilities
The facilities, technologies and processes to deliver end-to-end manufacturing.
Value-Add Capabilities
The team, expertise, and ingenuity to drive tangible added-value for our customers.
Devices
Extend capacity & reduce costs
Tap into our passion for innovation and we’ll help advance quality, extend your capacity and cut production costs.
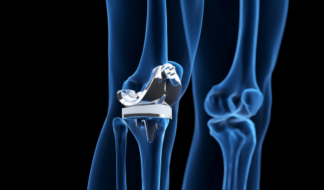
Knee Implants
Femoral implants, patella and tibia implants, and limb salvage.
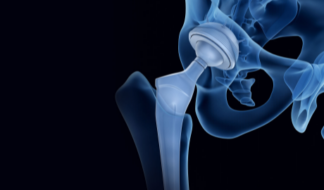
Hip Implants
Hip stems, acetabular cups, femoral heads and femoral liners.
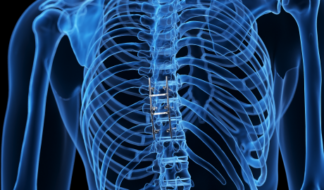
Spine Implants
Interbody cages, pedicle screws and rods, and SI fixation screws.
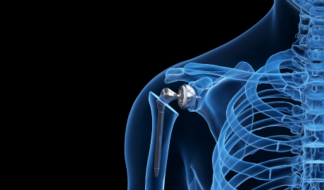
Trauma & Extremities
Shoulder & elbow, foot & ankle, tibia & femur, hand & wrist, cranial & maxofacial.


3D Printed Tantalum: A next-generation material for orthopedics?
Watch the webinar on-demand here: https://youtu.be/-PIIkfxG5XU?si=uogjrdcvCMHATApy Stay Ahead of the Curve in Orthopedic Innovation with Croom Medical’s Upcoming Webinar in collaboration with Global...

Croom Medical Wins Irish Medtech Partner/Supplier of the Year Award 2023
Croom Medical, a leading contract manufacturer of orthopedic implants and medical devices, has been awarded the prestigious Medtech Partner/Supplier of the Year Award at the ...
